
Salt-Organic Cocrystals as Solid Electrolytes
In an NSF-funded project, the Zdilla lab is working in collaboration with the Wunder group at Temple University on the generation and physical property measurement of salt-organic cocrystals as possible solid electrolyte materials for lithium ion batteries. Lithium ion batteries represent the state-of-the-art battery technology for rechargeability and energy density. Unfortunately, high-capacity batteries still rely on the use of flammable liquid electrolytes for the conduction of ions between the electrodes. This has led to fires and explosions. This is an increasing concern as humanity moves toward more energy-efficient vehicles that rely heavily on batteries as a power source.

Figure 1. Hangzhou taxi, which burst into flames, resulting in the city halting the use of electric taxis. Source: China Auto Web.
Significant research is going into the fabrication of solid electrolytes to replace flamable liquid electrolytes. The most explored classes are ceramic electrolytes (which have excellent conductivities, but poor adhesion to electrodes, and are difficult to fabricate into devices due to brittleness) and polymer electrolytes (which are soft solids, and maleable, but tend to have insufficient room-temperature conductivity).
We are currently exploring a new, simple type of materials as solid electrolytes, salt/organic cocrystals. Via specific crystallization protocols, we are able to prepare co-crystalline solids containing ions in low affinity chanels. This permits facile conduction of ions through the solid matrix.
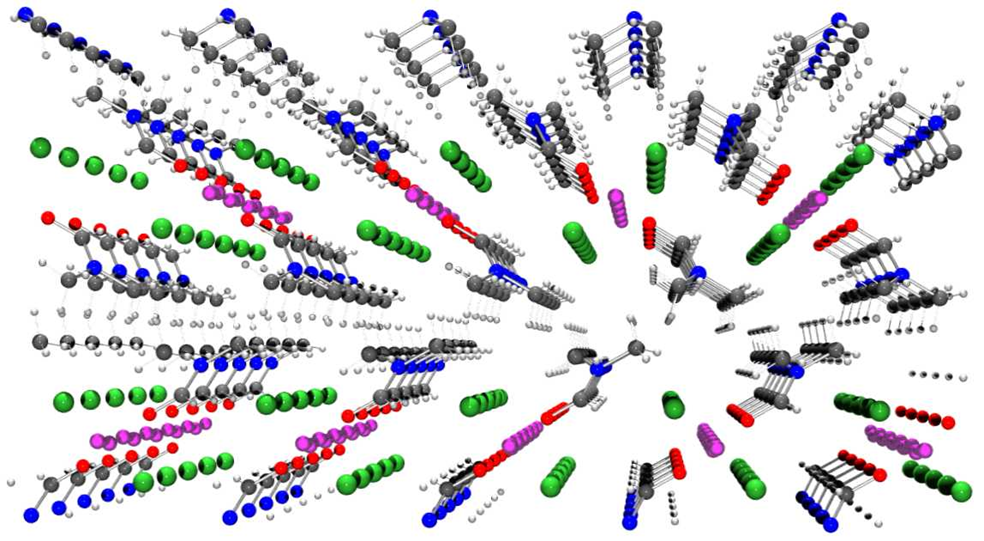
Figure 1. Crystal packing diagram of a dimethylformamide-lithium salt cocrystal showing channels of ions.
We are using single-crystal X-ray diffraction to characterize these materials. Electrochemical measurements carried out in the Wunder group demonstrate that our prototype materials show excellent conductivity, especially at low temperature. Powder diffraction to demonstrate that the materials' structures are maintained throughout the conductivity experiement.
The research in this area is currently expanding into materials with better thermal stability and higher melting points.
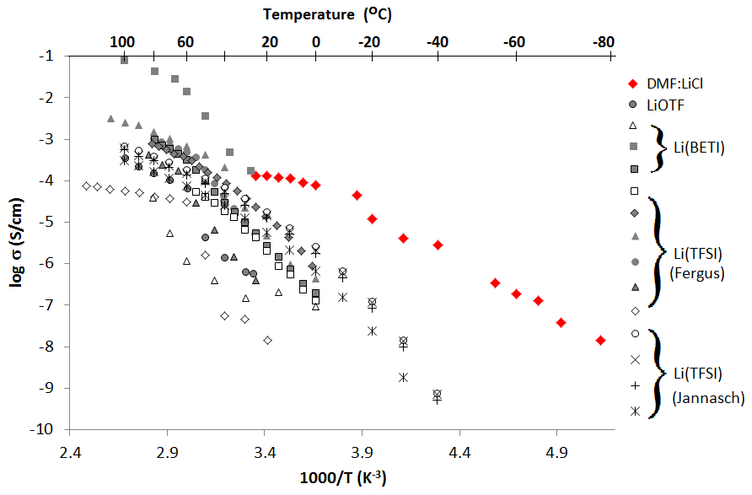
Figure 2. Conductivity measurements of two salt-organic cocrystals prepared in our group in comparison to pure polymer electrolytes. Figure reproduced from the Journal of Power Sources.[1]
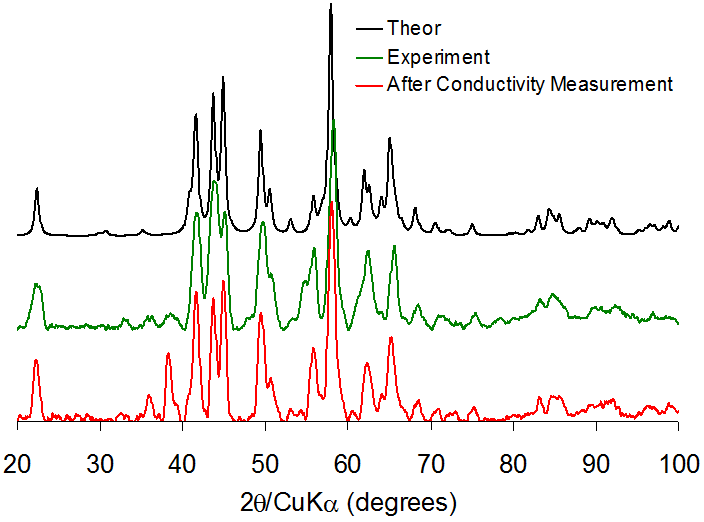
Figure 3. Powder diffraction patterns for a pyridine-based solid-state lithium electrolyte before and after electrochemical measurements, demonstrating the maintenance of the crystaline structure. Theoretical powder pattern based upon the single-crystal X-ray structure.
References
[1] Fergus, J. W. Journal of Power Sources 2010, 4554.
Selected Publications
Prakash, P; Fall, B.; Aguirre, J.; Sonnenberg, L. A.; Chinnam, P. R.; Chereddy, S.; Dikin, D. A.; Venkatnathan, A.; Wunder, S. L.; Zdilla, M. J. A soft co-crystalline solid electrolyte for lithium-ion batteries.Nature Mater. 2023. 22, 627–635.
Sonnenberg, L. A.; Paul, S. C.; Wunder, S. L.; Zdilla, M. J. The High-Temperature Polymorph of LiBF4. J. Phys. Chem. Lett. 2024, 15, 782–784.
Prakash, P.; Shylendran, A.; Fall, B.; Zdilla, M. J.; Wunder, S. L.; Venkatnathan, A. Mechanism of Ion Conduction and Dynamics in Tris(N,N-dimethylformamide) Perchloratosodium Solid Electrolytes. J. Phys. Chem. C. 2022, 126, 4744–4750.
Prakash, P. Ardhra, S Fall, B; Zdilla, M.J.; Wunder, S. L.; Venkatnathan, A. Solvate sponge crystals of (DMF)3NaClO4: reversible pressure/temperature controlled juicing in a melt/press-castable sodium-ion conductor. Chem. Sci. 2021, 12, 5574-5581.
Prakash, P. Ardhra, S Fall, B; Zdilla, M.J.; Wunder, S. L.; Venkatnathan, A. Solvate sponge crystals of (DMF)3NaClO4: reversible pressure/temperature controlled juicing in a melt/press-castable sodium-ion conductor. Chem. Sci. Advance Article
Fall, B.; Prakash, P.; Gau, M. R.; Wunder, S. L.; Venkatnathan, A.; Zdilla, M. J. Experimental and Theoretical Investigation of the Ion Conduction Mechanism of Tris (adiponitrile) perchloratosodium, a Self-Binding, Melt-Castable Crystalline Sodium Electrolyte. Chem. Mater. 2019. 31, 8850-8863.
Prakash, P.; Aguirre, J.; Van Vliet, M. M.; Chinnam. P. R.; Dikin, D. A.; Zdilla, M. J.; Wunder, S. L.; Venkatnathan, A. “Unravelling the structural and dynamical complexity of the equilibrium liquid grain-binding layer in highly conductive organic crystalline electrolytes.” J. Mater. Chem. A, 2018, 6, 4394-4404.
Fall, B; Jalil, A; Gau, M.; Chereddy, S.; Zdilla, M. J.; Wunder, S. L.; Chinnam, P. R. “Crystal structure and ionic conductivity of the soft solid crystal: isoquinoline •(LiCl).” Ionics. 2017, 24, 343-349.
Jalil, A.; Clymer, R. N.; Hamilton, C. R.; Vaddypally, S.; Gau, M. R.; Zdilla, M. J. “Structure of salts of lithium chloride and lithium hexafluorophosphate as solvates with pyridine and vinylpyridine and structural comparisons: (C5H5N)LiPF6, [p-(CH2 CH)C5H5N]LiPF6, [(C5H5N)LiCl]n, and [p-(CH2 CH)C5H5N]2-Li(μ-Cl)2Li[p-(CH2 CH)C5H5N]2” Acta Cryst. C. 2017, 73, 264-269.
Chinnam, P. R.; Fall, B.; Dikin, D. A.; Jalil, A.; Hamilton, C. R.; Wunder, S. L.; Zdilla, M. J. “A Self-Binding, Melt-Castable, Crystalline Organic Electrolyte for Sodium Ion Conduction.” Angew. Chem. Int. Ed. 2016, 55, 15254-15257.
Parameswara R. Chinnam, Rebecca N. Clymer, Abdel Aziz Jalil, Stephanie L. Wunder, and Michael J. Zdilla. “Bulk-phase, ion conduction in cocrystalline LiCl·N,N-dimethylformamide: A new paradigm for solid electrolytes based upon the Pearson Hard-Soft Acid-Base concept.” Chem. Mater. 2015. 27, 5479−5482

