
Bioinorganic model metalloclusters to mimic the oxygen evolving complex of Photosystem II.
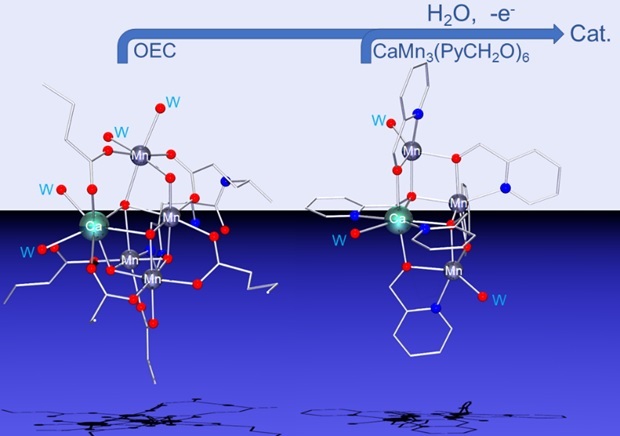 Figure 1. Left: Oxygen evolving complex of PSII. Right, synthetic model hemicubane cluster catalyzing water activation in electrocatalytic solvent oxidation.
Figure 1. Left: Oxygen evolving complex of PSII. Right, synthetic model hemicubane cluster catalyzing water activation in electrocatalytic solvent oxidation.
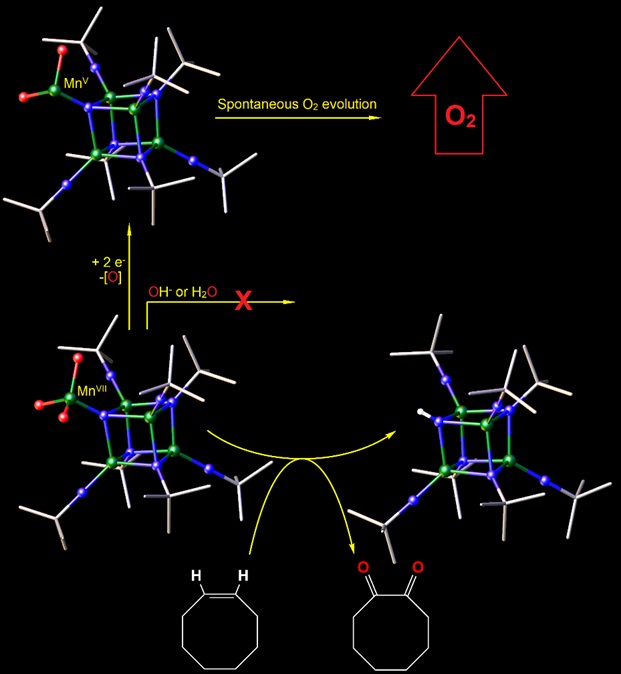 Figure 2. Bottom: Model cubane compound featuring MnVII=O moieties do oxidize alkenes, but do not evolve O2.8 Top: Reduction of the pendant moiety to MnV results in spontaneous O2 evolution.
Figure 2. Bottom: Model cubane compound featuring MnVII=O moieties do oxidize alkenes, but do not evolve O2.8 Top: Reduction of the pendant moiety to MnV results in spontaneous O2 evolution.
Photosystem II is the origin enzyme for the electron transport chain oxygenic photosynthesis, taking low-energy electrons from water, exciting them with light, and using them to create cellular fuel, and evolving O2 as the oxidized byproduct. An understanding of the functioning of the oxygen evolving complex (OEC, See Figure 1 left) of this enzyme has been elusive. Though much knowledge has been gleaned from XFEL, X-ray spectroscopy, biophysical, and computational studies, there remains much disagreement on the mechanism of oxygen evolution.[1] Model complex chemistry is uniquely poised among methods of bioinorganic mechanistic inquiry to probe proposals of reaction mechanism. And while model complex can never prove the mode of action of an enzyme, accurate and reactive model compounds can help to support (or disprove) certain types of proposed mechanisms. Two examples of our findings are described here.
We have described a series of synthetic “hemicuboidal” clusters (meaning that they are cube-shaped clusters like the OEC, but missing a corner metal atom). These clusters varied in composition and geometry. We found that the cluster most similar to in structure (Ca-Mn cuboid with a “dangling” Mn) and composition (one calcium ion and three manganese ions) showed the best electrocatalytic activity in activating water to participate in oxidation of the electrochemically stable solvent propylene carbonate (Figure 1).[2]
In a second example, we prepared the first high-oxidation-state Mn cubane cluster featuring a pendant Mn=O moiety similar to that proposed by some for the “nucleophilic attack mechanism” for OEC turnover. Our compound “stacks the deck” for a nucleophilic attack on the electron-poor terminal oxo ligand by water or hydroxide by virtue of the neighboring MnVII metal (more Lewis acidic than Nature’s formal MnV). However, our MnVII=O is unreactive toward two-electron nucleophiles, though it is active in the oxidation of alkenes,[3] it does not show biomimetic Oxygen Evolution, suggesting there is more to the mechanism than a highly electrophilic oxygen.
Other examples of our bio-inspired cluster chemistry are given in the references list below.[1-18]
References
(1) Alec Bigness; Shivaiah Vaddypally; Zdilla, M. J.; Mendoza-Cortes, J. L. Ubiquity of Cubanes in Bioinorganic Relevant Compounds. Coord. Chem. Rev. 2021, 450, 214168.
(2) Koellner, C. A.; Gau, M. R.; Polyak, A.; Bayana, M.; Zdilla, M. J. Hemicubane topological analogs of the oxygen-evolving complex of photosystem II mediating water-assisted propylene carbonate oxidation. Chem. Commun. 2022, 58, 2532-2535.
(3) Vaddypally, S.; Kondaveeti, S. K.; Karki, S.; VanVliet, M. M.; Levis, R. J.; Zdilla, M. J. Reactive Pendant Mn=O in a Synthetic Structural Model of a Proposed S4 State in the Photosynthetic Oxygen Evolving Complex. J. Am. Chem. Soc. 2017, 139, 4675-4681
(4) Hamilton, C. R.; Zdilla, M. J. Structure of a pentamanganese(II)-phenoxide cluster with a central five-coordinate oxide: MnII5([mu]-OPh)6([mu]3-OPh)2([mu]5-O)(Py)6.Py (Py is pyridine). Acta Crystallographica Section C 2017, 73, 270-275.
(5) Gau, M. R.; Hamilton, C. R.; Zdilla, M. J. Preparation of a “twisted basket” Mn4N8 cluster: a two-hydrogen-atom reduced analogue of the Mn4N8 pinned butterfly. Chem. Commun. 2014, 50, 7780-7782
(6) Vaddypally, S.; Kondaveeti, S. K.; Roudebush, J. H.; Cava, R. J.; Zdilla, M. J. Formation of the tetranuclear, tetrakis-terminal-imido Mn4IV(NtBu)8 cubane cluster by four-electron reductive elimination of tBuN=NtBu. The role of the s-block ion in stabilization of high-oxidation state intermediates. Chem. Commun. 2014, 50, 1061-1063.
(7) Vaddypally, S.; Kondaveeti, S. K.; Zdilla, M. J. An isolable, metastable, geometrically unique manganese(iv) trihydrazide complex poised for reactivity. Chem. Commun. 2011, 47, 9696-9698.
(8) Zdilla, M. J. 5.04 Electronic Structure of Paramagnetic Iron and Manganese Cluster Compounds: Historical Developments and Current Understanding. In Comprehensive Coordination Chemistry III; Constable, E. C., Parkin, G., Que Jr, L., Eds.; Elsevier: Oxford, 2021, p 45-81.
(9) Kondaveeti, S. K.; Vaddypally, S.; McCall, J. D.; Zdilla, M. J. Electronic structure and solution behavior of a tris(N,N'-diphenylhydrazido)manganese(iv) propeller complex. Dalton Transactions 2012, 41, 8093-8097.
(10) Gau, M. R.; Zdilla, M. J. Multinuclear Clusters of Manganese and Calcium with Polyhedral Oligomeric Silsesquioxane ligands: Synthesis and Dioxygen Activation for Si-O Cleavage Reactivity. Inorg. Chem. 2020, 60, 2866–2871.
(11) Kondaveeti, S. K.; Vaddypally, S.; Lam, C.; Hirai, D.; Ni, N.; Cava, R. J.; Zdilla, M. J. Synthesis, Structure, and Magnetic Studies of Manganese–Oxygen Clusters of Reduced Coordination Number, Featuring an Unchelated, 5-Coordinate Octanuclear Manganese Cluster with Water-Derived Oxo Ligands. Inorg. Chem. 2012, 51, 10095-10104.
(12) Vaddypally, S.; Kondaveeti, S. K.; Zdilla, M. J. Synthesis of a High-Valent, Four-Coordinate Manganese Cubane Cluster with a Pendant Mn Atom: Photosystem II-Inspired Manganese–Nitrogen Clusters. Inorg. Chem. 2012, 51, 3950-3952.
(13) Vaddypally, S.; Xu, C.; Zhao, S.; Fan, Y.; Schafmeister, C. E.; Zdilla, M. J. Architectural Spiroligomers Designed for Binuclear Metal Complex Templating. Inorg. Chem. 2013, 52, 6457-6463.
(14) McCall, J. D.; Vaddypally, S.; Kondaveeti, S. K.; Zdilla, M. J. Easy access to the Wilkinson tris(tert-butylimido)nitridomanganate(VII) complex from commercially available starting materials. Inorg. Chem. Commun. 2013, 37, 225-227.
(15) Hamilton, C. R.; Baglia, R. A.; Gordon, A. D.; Zdilla, M. J. Synthesis of Tetranuclear, Four-Coordinate Manganese Clusters with “Pinned Butterfly” Geometry Formed by Metal-Mediated N−N Bond Cleavage in Diphenylhydrazine. J. Am. Chem. Soc. 2011, 133, 4208-4211.
(16) Hamilton, C. R.; Gau, M. R.; Baglia, R. A.; McWilliams, S. F.; Zdilla, M. J. Mechanistic Elucidation of the Stepwise Formation of a Tetranuclear Manganese Pinned Butterfly Cluster via N–N Bond Cleavage, Hydrogen Atom Transfer, and Cluster Rearrangement. J. Am. Chem. Soc. 2014, 136, 17974-17986.
(17) Vaddypally, S.; Tomlinson, W.; O’Sullivan, O. T.; Ding, R.; Van Vliet, M. M.; Wayland, B. B.; Hooper, J. P.; Zdilla, M. J. Activation of C–H, N–H, and O–H Bonds via Proton-Coupled Electron Transfer to a Mn(III) Complex of Redox-Noninnocent Octaazacyclotetradecadiene, a Catenated-Nitrogen Macrocyclic Ligand. J. Am. Chem. Soc. 2019, 141, 5699-5709.
(18) Gau, M. R.; Zdilla, M. J. A Protocol for Safe Lithiation Reactions Using Organolithium Reagents. Journal of Visualized Experiments 2016, 117, e54705.

