
Metal-Nitrogen Based High Energy Density Materials
The military has an interest in high energy materials (HEDM) with increased shattering potential for focused target strikes with decreased collateral damage. To achieve this end, the generation of disruptive energetic materials (~10x the shattering potential of RDX) is a goal.
In order to breach the theoretical ceiling for energy density in CHNO-based explosives, the inclusion of new atoms is necessary. The inspriation for this project came originally from an inorganic chemist's inspection of the instability of an organic explosive, RDX, the main ingredient in plastic explosive:
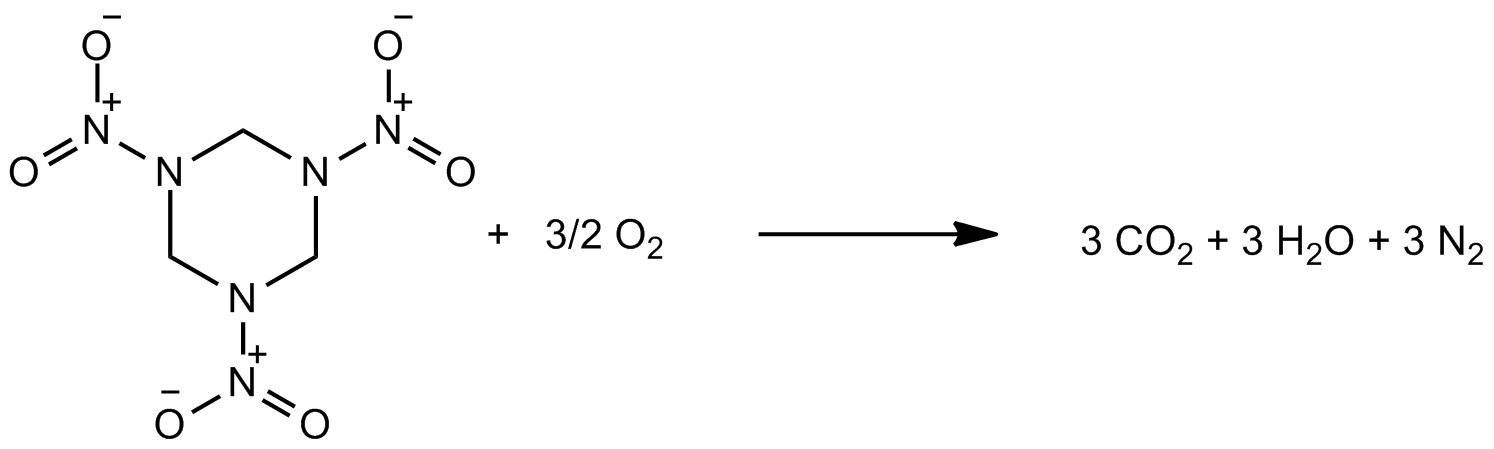
Traditionally, an organic synthetic chemist might say that RDX is high in energy due to the presence of high energy bonds, which are traded for low energy bonds in the product carbon dioxide, water, and dinitrogen, and the remaining energy is released as heat and pressure-volume work (an explosion). While this view is reasonable, an equally valid view of this molecule involves a consideration of the oxidation states of the atoms: that the RDX molecule posesses atoms in a "redox-frustrated" arrangement, with low-oxidation state atoms (carbon, amine-nitrogen) in close proximity to high-oxidation state atoms (nitro-nitrogens), and that the release of the energy occurs upon atoms reaching their most stable oxidation states in the products, liberating heat and work.
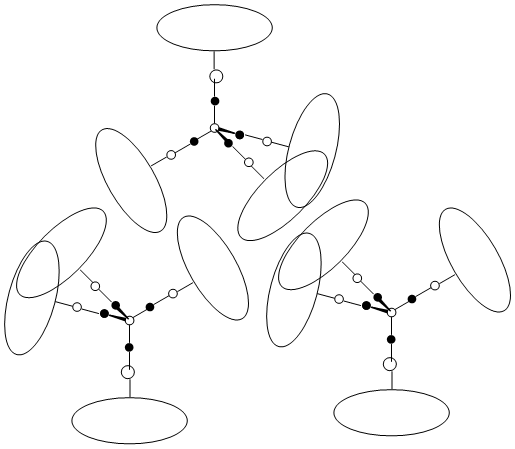
Figure 1. Design concept of a Mn-based HEDM, with central high-oxidation state manganese atoms surrounded by nitrogen-rich spacers, combining to give a redox-frustrated molecule with low lattice energy.
This view of a HEDM spurs new ideas as to how to increase energy density. The inclusion of functionality with larger oxidizing power than nitro groups would be expected to increase the energy density by a significant amount. An obvious choice of atom for this purpose is manganese, which can exist in a number of high oxidation states, (IV-VII). For instance, permanganate ion (MnO4-) is one of the strongest oxidants in nature (far stronger than a nitro group). Inclusion of such species into nitrogen-rich ligand frameworks would provide reducing fuel atoms (organic carbon and reduced nitrogen) like in RDX, but would replace the oxidizer component, nitro groups, with a much stronger oxidant, resulting in more liberated energy upon decomposition.
This view of a HEDM spurs new ideas as to how to increase energy density. The inclusion of functionality with larger oxidizing power than nitro groups would be expected to increase the energy density by a significant amount. An obvious choice of atom for this purpose is manganese, which can exist in a number of high oxidation states, (IV-VII). For instance, permanganate ion (MnO4-) is one of the strongest oxidants in nature (far stronger than a nitro group). Inclusion of such species into nitrogen-rich ligand frameworks would provide reducing fuel atoms (organic carbon and reduced nitrogen) like in RDX, but would replace the oxidizer component, nitro groups, with a much stronger oxidant, resulting in more liberated energy upon decomposition.
To this end, our group is exploring the synthesis of nitrogen-rich complexes of high-oxidation state manganese. The design concept is described in figure 1, where high-oxidation state metal centers are ligated and separated by nitrogen-rich organic spacers, which act as fuel molecules.
 Figure 2. Structure of the manganese (VII) tris(imido)nitrido cluster originally reported by Wilkinson [1], for which we have developed an improved synthesis [2].
Figure 2. Structure of the manganese (VII) tris(imido)nitrido cluster originally reported by Wilkinson [1], for which we have developed an improved synthesis [2].
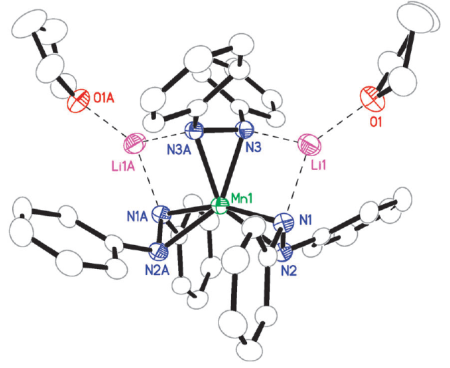 Figure 3. Structure of a Mn(IV) tris-hydrazide propeller complex [3]. This complex is redox frustrated, being composed of reducing hydrazide ligands and an oxidizing Mn(IV) ion.
Figure 3. Structure of a Mn(IV) tris-hydrazide propeller complex [3]. This complex is redox frustrated, being composed of reducing hydrazide ligands and an oxidizing Mn(IV) ion.
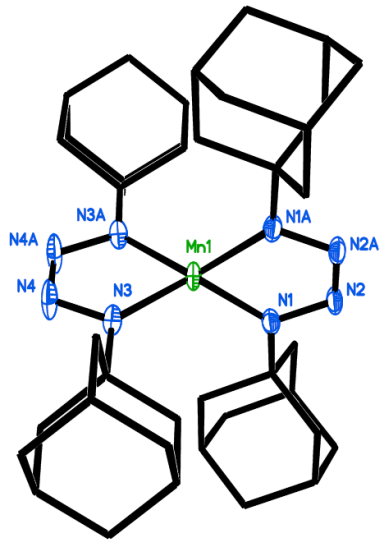
Figure 4. Structure of a formal Mn(IV) tetrazene ligand system. In actuallity, the system behaves as a Mn(II) with two redox-non-innocent anion radical tetrazene ligands.
Among the ligand motifs being explored are single-nitrogen ligands (nitride, imide, amide) as well as oligo-nitrogen ligands including hydrazine and tetrazene. Among our first contributions to these materials was the development of a protocol for the synthesis of the [Mn(VII)(N)(t-BuN)3]2- ion (Fig. 2), a nitrogen analouge of permangenate [1] from commercially available starting materials. [2]
We have reported the synthesis [3] and electronic structure ans solution behavior [4] of a geometrically unique manganese tris-hydrazide molecule (Fig 3). This molecule is redox frustrated in that it contains reducing diphenyl hydrazide ligands directly bonded to and oxidizing Mn(IV) ion. The compound is quasi-stable and decomposes under certain conditions, relaxing to Mn(II).
Most recently, we have begun work on manganese tetrazene systems. Tetrazene (or tetraazadiene) is a N4 ligand system that exhibits redox non-innocent behavior, and analagous to the common alpha-diimine ligand, can exist in three oxidation states: Neutral, anion radical, and dianion. The attachment of this ligand to an oxidizing formal Mn(IV) actually results in partial oxidation of the ligand to the anion radical state with a manganese (II) ion.
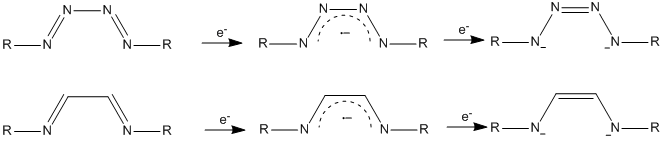
Figure 5. Redox-non-innocent behavior of tetrazene ligand in comparison to the more common alpha-diimine ligand
We are currently working to expland this class of compounds to ones with higher nitrogen content in the R-groups or smaller-alkyl systems to increase the overall nitrogen content.
References
[1] Danopoulos, A. A.; Wilkinson, G.; Sweet, T. K. N.; Hursthouse, M. B., tert-Butylimido compounds of manganese-(VII), -(VI), -(V) and -(II); nitrido, amido, alkyl, zinc and aluminium compounds. Journal of the Chemical Society, Dalton Transactions 1995, 205-216.
[2] McCall, J. D.; Vaddypally, S.; Kondaveeti, S. K.; Zdilla, M. J., Easy access to the Wilkinson tris(tert-butylimido)nitridomanganate(VII) complex from commercially available starting materials. Inorganic Chemistry Communications 2013, 37, 225-227.
[3] Vaddypally, S.; Kondaveeti, S. K.; Zdilla, M. J., An isolable, metastable, geometrically unique manganese(iv) trihydrazide complex poised for reactivity. Chemical Communications 2011, 47 (34), 9696-9698.
[4] Kondaveeti, S. K.; Vaddypally, S.; McCall, J. D.; Zdilla, M. J., Electronic structure and solution behavior of a tris(N,N[prime or minute]-diphenylhydrazido)manganese(iv) propeller complex. Dalton Transactions 2012, 41 (26), 8093-8097.
Selected Publications
Allen, J. E.; Zybin, S. V.; Morozov, S. I.; O’Sullivan, O. T.; Kawamura, C.; Waxler, D. E.; Hooper, J. P.; Goddard III, W. A.; Zdilla, M. J. J. Am. Chem. Soc. 2024, 146, 4500–4507.
Vaddypally, S.; Byrne, A. N.; Goldsmith, C. F.; Zdilla, M. J.; Kiselev, V. G. Metal-Free Reversible Double Cyclization of Cyanuric Diazide to an Asymmetric Bitetrazolate via Cleavage of the Six-Membered Aromatic Ring. Inorg. Chem. 2024, 63, 2322–2326.
Bacon, A. M.; Tomlinson, W.; Hooper, J. P.; Zdilla, M. J. Titanium(II) as a Fuel Atom in Energetic Materials. Inorg. Chem. 2023, 62, 9285–9290.
Zybin, S. V.; Morozov, S. I.; Prakash, P.; Zdilla, M. J.; Goddard, W. A. Reaction Mechanism and Energetics of Decomposition of Tetrakis(1,3-dimethyltetrazol-5-imidoperchloratomanganese(II)) from Quantum-Mechanics-based Reactive Dynamics. J. Am. Chem. Soc. 2021, 143, 16960-16975.
Vaddypally, S.; Kiselev, V. G.; Byrne, A. N.; Goldsmith, F. C.; Zdilla, M. J. Transition-metal-mediated reduction and reversible double-cyclization of cyanuric triazide to an asymmetric bitetrazolate involving cleavage of the six-membered aromatic ring. Chem. Sci. 2021. 12, 2268-2275.
O’Sullivan, O. T.; Zdilla, M. J. Properties and Promise of Catenated Nitrogen Systems as High-Energy-Density Materials. Chem. Rev. 2020. 120, 5682–5744.
Vaddypally, S.; Tomlinson, W.; O'Sullivan, O. T.; Ding, R.; Van Vliet, M; Wayland, B. B.; Hooper, J. P.; Zdilla, M. J. Activation of CH, NH, and OH bonds via proton-coupled electron transfer to a Mn (III) complex of redox-noninnocent octaazacyclotetradecadiene, a catenated-nitrogen macrocyclic ligand. J. Am. Chem. Soc. 2019, 141, 5699-5709.)
Lundell, C. E.; O’Sullivan, O. T.; Gau, M. R.; Zdilla, M. J. “Syntheis of Two Lead Complexes of Propellant Stabilizer Compounds: In Pursuit of Novel Propellant Additives.” ChemistrySelect 2017, 2, 11673 –11676.
O’Sullivan, O. T.; Zdilla, M. J. “Manganese-mediated linkage of perchlorate to aminotetrazoles gives twice the energy density of the unmetallated salt.” Chem. Eur. J. 2017, 23, 14138-14142.
Vaddypally, S.; McKendry, I. G.; Tomlinson, W.; Hooper, J. P.; Zdilla, M. J. “Electronic structure of manganese complexes of the redox-non-innocent tetrazene ligand and evidence for the metal-azide/imido cycloaddition intermediate.” Chem. Eur. J. 2016, 22,10548 –10557
Kondaveeti, S. K.; Vaddypally, S.; McCall, J. D.; Zdilla, M. J. “Electronic structure and solution behavior of a tris(N,N′-diphenylhydrazido)manganese(IV) propeller complex.” Dalton Trans., 2012, 41, 8093.
Vaddypally, S.; Kondaveeti, S. K.; Zdilla, M. J. “An isolable, metastable, geometrically unique manganese(IV) trihydrazide complex poised for reactivity.” Chem. Commun. 2011, 47, 9696.

